What is (6)-Gingerol?
What is it? (6)-Gingerol is one of the main phenolic compounds responsible for the characteristic pungent sensation of ginger, obtained from the root of ginger (Zingiber officinale). Because it carries a "vanilloid"-like core, its biological effects are being investigated through its interaction with some sensory receptors.
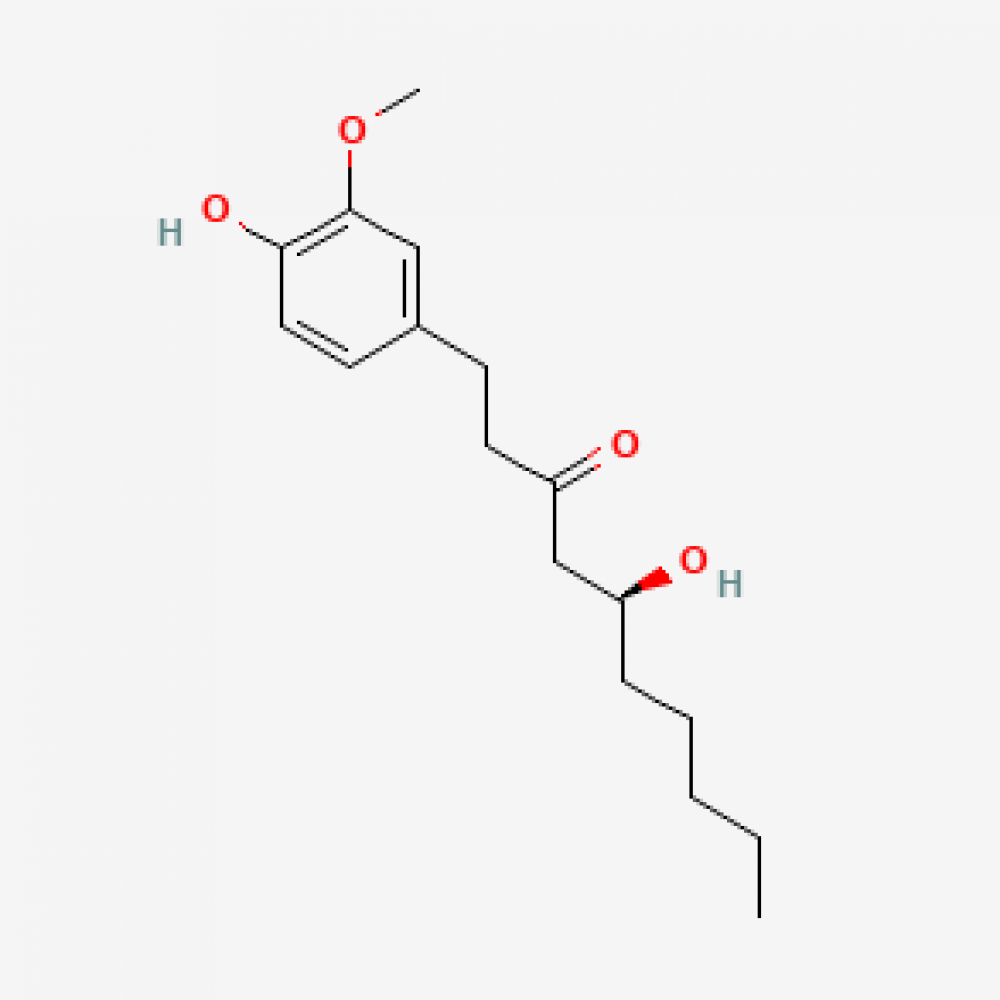
Structure
It contains hydroxy + methoxy substitutions (vanilloid motif) in the aromatic ring and hydroxy-ketone functionalities in the aliphatic chain. This structure shapes both its membrane interaction/solubility behavior and its potential to bind to some target proteins. The basic structural identifiers are given below from the PubChemLite record.
Molecular Identification Card
• Molecular Formula: C₁₇H₂₆O₄
• Molecular Weight: 294.4 g/mol
• PubChem CID: 442793
• PubChem SID: 135650304 (An example of a reported SID for gingerol in PubChem; other SIDs may be found in different supplies/contexts.)
• CAS Number: 23513-14-6
• Synonyms: 6-Gingerol, [6]-Gingerol, Gingerol, (+)-6-gingerol, Geijerol (common uses)
Structural Identifiers:
• SMILES: CCCCCC@@HO
• InChIKey: NLDDIKRKFXEWBK-AWEZNQCLSA-N
• IUPAC Name: (5S)-5-hydroxy-1-(4-hydroxy-3-methoxyphenyl)decan-3-one
MeSH Pharmacological Classification
(6)-Gingerol is not a molecule standardized/linked to ATC as a drug active ingredient in the clinic; it is mostly classified as a natural product/phytoactive in the literature and is indexed in PubMed in studies in the context of the chemical "gingerol". (Therefore, it is not possible to give a single official drug class under the heading "MeSH pharmacological class" in most sources.)
ATC Code
None / not assigned (no official ATC classification as a drug).
Mechanism of Action
The mechanism for (6)-gingerol is considered as a sum of effects investigated across different biological axes, rather than an indication-specific “single target” approach:
• TRPV1 (vanilloid receptor) activation
It has been shown that ginger-derived vanilloid compounds create a “pungent” sensation by activating TRPV1, and the ligand-channel interaction has been investigated at the structural level.
• Investigations of effects via pain-related ion channels (e.g., Na⁺ channels)
There are preclinical studies in which [6]-gingerol and [6]-shogaol have been investigated via sodium channel-related mechanisms in specific pain models.
• Anti-inflammatory/antioxidant axis (multiple pathways)
Anti-inflammatory effects have been investigated in different disease models (e.g., intestinal inflammation), particularly through markers of inflammation and oxidative stress.
The mechanism literature is extensive; Target/pathway-level inferences are highly sensitive to model, dose, and formulation.
Other Additions and Synergistic Effects
From a formulation perspective, (6)-gingerol is considered together with components from the same “ginger phenolics” family:
• 6-Shogaol / 6-Gingerol association: They are mentioned together in the bioactive profile of ginger; the “component community” approach is common in the axis of immunomodulation and inflammation.
• Vanilloid-focused sensory effect: The sensory profile associated with TRPV1 activation also determines the “perceptual effect” side (pain/burning) in product design.
Research
Examples of scientific studies conducted with 6-gingerol;
• A study investigating the effects of 6-gingerol on inflammation and disease severity indicators in a DSS-induced colitis model.
• Mechanistic/structural study on TRPV1 activation and binding of vanilloid compounds.
• A study investigating the effects of 6-gingerol and 6-shogaol on pain mechanisms and ion channel-related effects.
• Reviews on the immunomodulatory potential of gingerol/ginger components (particularly 6-gingerol and 6-shogaol) are available.
For more information about products containing 6-gingerol developed at the CT Pharma Research R&D Laboratory, please check the link!
Referances
• https://pubchemlite.lcsb.uni.lu/e/compound/442793
• https://drugmap.idrblab.net/data/drug/details/DMNXYSM
• https://pmc.ncbi.nlm.nih.gov/articles/PMC6692589/
• https://onlinelibrary.wiley.com/doi/10.1002/ptr.5286